Determination of microbial biomass in soils
TOC in soil science

Fertile soils contain a multitude of microorganisms. They are responsible for the degradation of organic substances and protection of the nutrient cycle. To evaluate soils in terms of their biodegradability and their fertility, the microbial biomass of the soil, i.e. the organic carbon fraction that is bound to these tiniest organisms, is determined.
The fumigation-extraction method is a commonly used method for the determination of microbial biomass in soils. The microbial biomass is described regarding extractable organic carbon compounds before and after killing off the microorganisms.
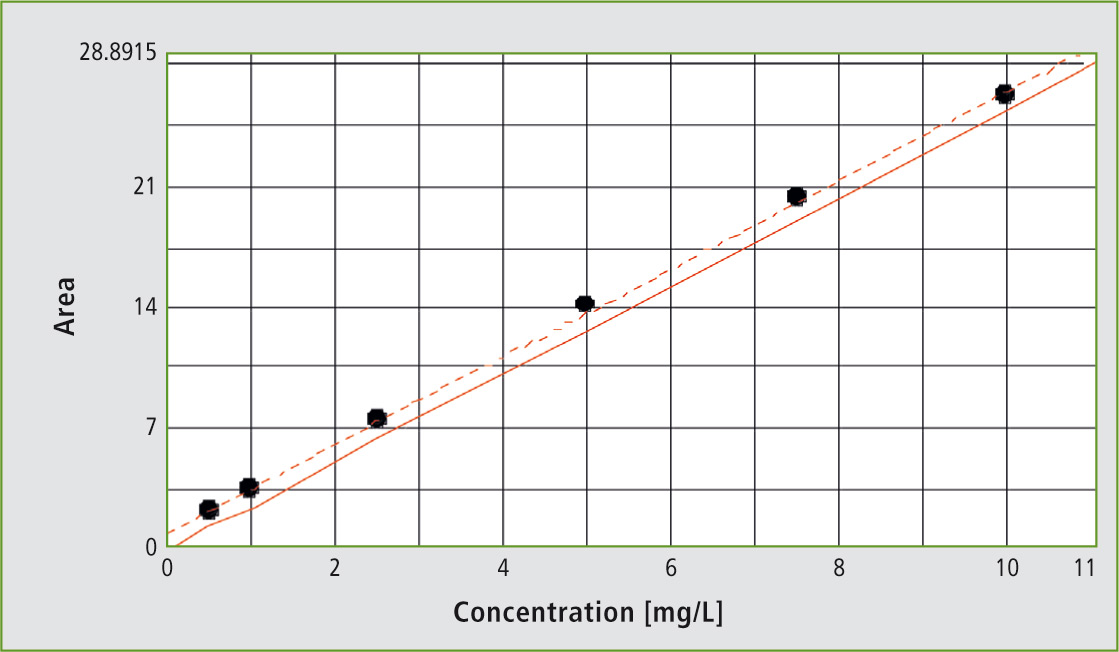 Figure 1: Automated NPOC calibration in the range of 1 to 10 mg/L
Figure 1: Automated NPOC calibration in the range of 1 to 10 mg/L
In the fumigation-extraction method, a subsample of the soil is treated with chloroform gas in a suitable apparatus (e.g. a desiccator) over an extended period of time of at least 24 hours. This destroys the cell walls and kills off the microorganisms. After fumigation, the chloroform remaining in the soil is removed.
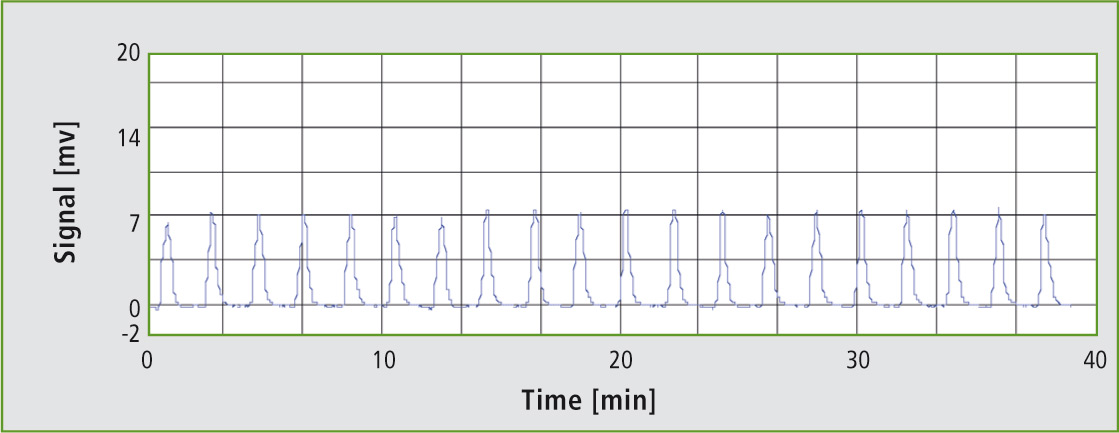 Figure 2: Injection 1 – 20
Figure 2: Injection 1 – 20
DOC is determined first
Subsequent to fumigation, each fumigated subsample and one non-fumigated soil sample is mixed with a 0.5 M (mol/L) potassium sulfate solution and then shaken. After filtration of the eluate, the DOC (dissolved organic carbon) of the extracts is determined. Since experience has shown, not all cells are destroyed and extracted, and an empirical correction factor is additionally applied. (An exact description of the fumigation-extraction method is found in the EN ISO 14240-2:2011 standard).
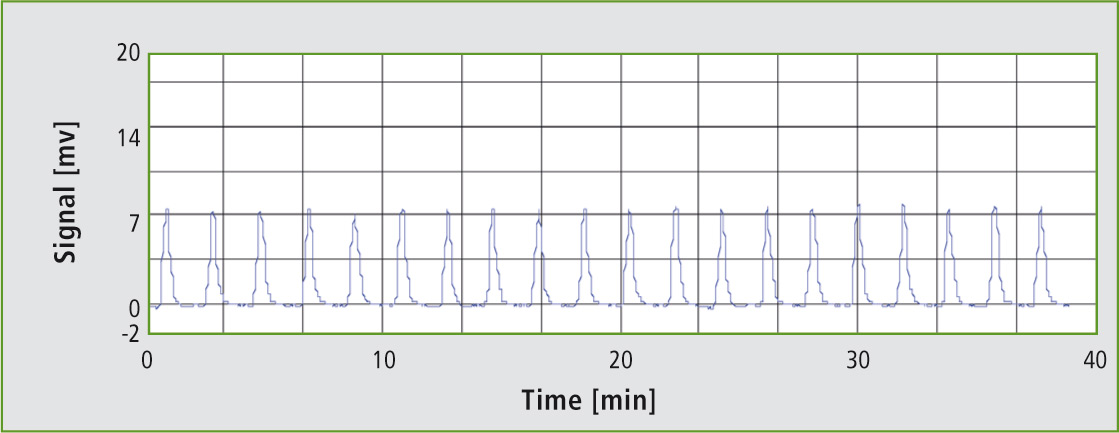 Figure 3: Injection 581 – 600
Figure 3: Injection 581 – 600
DOC analyses of the potassium sulfate extracts are carried out using a TOC analyzer. In this procedure, the extract is mixed with an acid to remove the inorganic carbon fraction, whereby carbonates and hydrogen carbonates are converted to CO2. The carbon dioxide is purged from the sample using a purging gas. An aliquot of the pretreated extract is subsequently injected onto a hot platinum catalyst, whereby the organic substances are oxidized to CO2. A carrier gas transfers the carbon dioxide to an NDIR detector, which detects the amount of CO2.
Kit for high salt loads
For many TOC analyzers, the high salt loads of the extracts can, however, cause problems. The extraction solution alone has a salt load of approximately 87 g/L. High salt loads can lead to clogging of the catalysts and the combustion tubes. When the combustion temperature is too high, the salts form a melt that blocks the active sites of the catalyst.
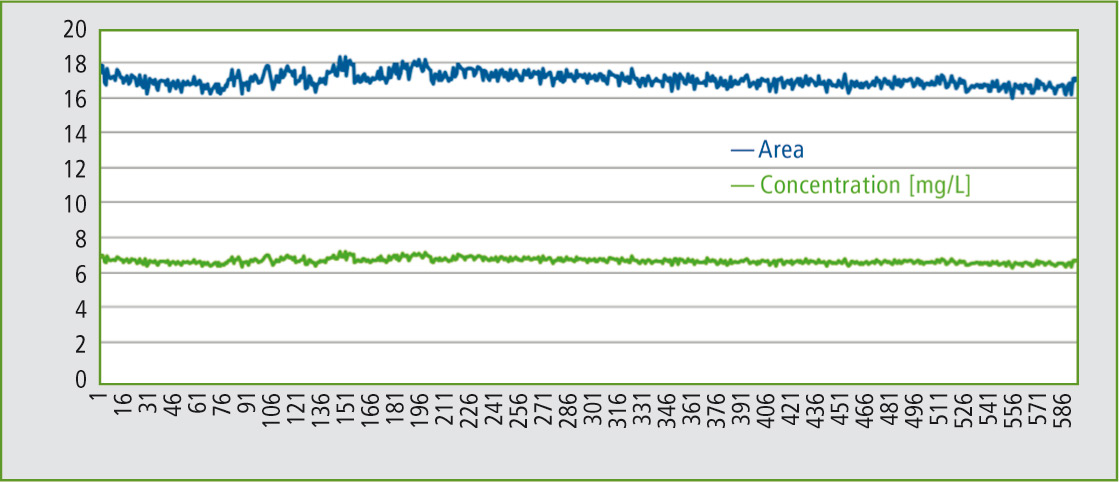 Figure 4: Sequence (area and concentration) of the 600 injections
Figure 4: Sequence (area and concentration) of the 600 injections
Therefore, Shimadzu’s TOC-L uses a catalytic combustion temperature of 680 °C, which is lower than the melting point of most salts. In addition, a special kit for high salt load samples is available for such measuring tasks. It consists of a combustion tube featuring a specific geometry and a unique catalyst mixture. This particular catalyst can handle up to 12 times more salt than a conventional catalyst.
Long-term test with 600 injections
A long-term test should show that an application, such as the analysis of soil extracts from a 0.5 M potassium sulfate solution can be carried out problem-free using a kit for high salt load samples. For this test, a 0.5 M K2SO4 solution (corresponding to 87 g/L) was injected 600 times onto the kit for high-salt samples.
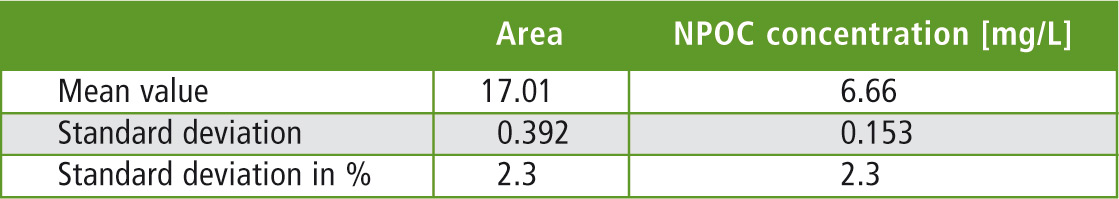 Table 1: Statistical evaluation of the 600 injections
Table 1: Statistical evaluation of the 600 injections
Method parameters
NPOC preparation: automated using the ISP module of the TOC-L analyzer
Acidification: 2 %
Sparging: 6 minutes
Injection volume: 50 µL
Calibration: 6-point calibration using the automated dilution function, from a stock solution in the range of 0.5 mg/L to 10 mg/L.
Results
Even after 600 injections, the peak shapes remain exactly the same as at the start of the injection series. The results remain stable over the entire time period. As one can see, the TOC-L in combination with the kit for high salt load samples is highly suitable for TOC determination of salt-containing samples, such as soil extracts from a 0.5 M potassium sulfate solution.