Food and food packaging: non-intentionally added substances
FTIR, EDX and LC-GC-online Technique for colored, non-transparent plastic packaging

Food can be contaminated not only through direct contact with its packaging, but also along the entire production and commercial chain. So far, legislation on the migration of substances from plastic packaging into food in particular has covered the starting materials used in the plastics manufacturing. Regarding health risks related to the transfer of substances, the so-called NIAS (non-intentionally added substances) should be focused on more strongly; these substances are added non-intentionally during manufacturing of plastic packaging materials. These can include impurities originating from raw materials, reaction and degradation products in manufacturing as well as impurities arising from transport and production [1].
Contamination during the production process
Possible sources for impurities can be found in the production process, including preservation and bottling, in which metals can migrate into food and beverages. Often, raw as well as processed products are in prolonged contact with materials such as stainless steel, copper, glass and other equipment.
Impurities in the packaging material
Another source is the packaging material. It is in close contact with the food – during the entire transport, storage in retail as well as in the consumer’s home.
Currently, there is much discussion on food packaging with saturated and aromatic mineral oil hydrocarbons (MOSH – mineral oil saturated hydrocarbons and MOAH – mineral oil aromatic hydrocarbons).
The MOSH fraction consists of linear and branched alkanes as well as alkyl substituted cycloalkanes, whereas the MOAH fraction consists of alkylated polyaromatic hydrocarbons with up to four aromatic rings. The main focus is on the aromatic fraction, which is suspected of being potentially carcinogenic and mutagenic [2]. The proportion of the aromatic fraction is approximately 15 – 30 % of the total mineral oil fraction.
In addition to the mineral oil fractions MOSH and MOAH, the so-called POSH (polyolefinic oligomeric saturated hydrocarbons) are focused, i.e. oligomers that can migrate from plastic packaging (PE, PP).
Scope of the new plastics regulation
In principle, the Commission Regulation (EU) No. 10/2011 on plastic materials and articles that come into contact with food stipulates that impurities as well as reaction and degradation products must be assessed by the manufacturer in accordance with internationally accepted scientific principles of risk management.
As of 1 January 2016, the amended test conditions described therein fully apply. In addition to overall migration (migration limit: 60 mg/kg food), there are many other specific migration limit values that must be adhered to.
‘Multi-material multi-layer materials’ are also included in the new plastics regulation as well as materials or objects consisting of two or more layers of different materials of which at least one consists of plastic [3]. As reported in the Shimadzu News 1/2017, a total of 32 transparent, colorless and 50 imprinted food packagings were investigated. This article now focuses on colored non-transparent plastic packagings. The plastics were analyzed using FTIR and EDX spectroscopy as well as an LC-GC online system. Spectroscopic methods offer the advantage of non-destructive sample analysis with little time expenditure. The chromatographic method can be fully automated.
Within the scope of the investigation, 43 samples of different origin were analyzed. This included, for instance, caps or closures of bottles and cups, vegetable nets and trays. First, the samples were identified using FTIR spectroscopy and later analyzed for non-intentionally added substances such as heavy metals and mineral oil hydrocarbons.
Identification of plastics using FTIR spectroscopy
All samples were analyzed in the first step using FTIR spectroscopy. This enabled a non-destructive investigation of surfaces in which the main components of the outer and inner surfaces of the plastic packagings could be identified. The absorption was measured using Shimadzu’s IR Tracer-100 equipped with a diamond ATR. In this method, the IR beam penetrates the sample to a depth of about 2 µm, whereby the intensity of the reflected light is attenuated with respect to the irradiated light.
In a second complementary step, an EDX analysis was executed using Shimadzu’s EDX-8000P. Upon suitable excitation, each element emits characteristic X-rays allowing the element’s identification. Using energy dispersive X-ray fluorescence analysis, elements from carbon up to uranium can easily be detected in the lower ppm range.
To analyze the samples, a total of 48 IR and 40 EDX spectra were recorded. Table 1 lists samples that include packagings consisting of one or two main polymers. Most of them were black or white, while otherswere blue, yellow, orange, green or gold.
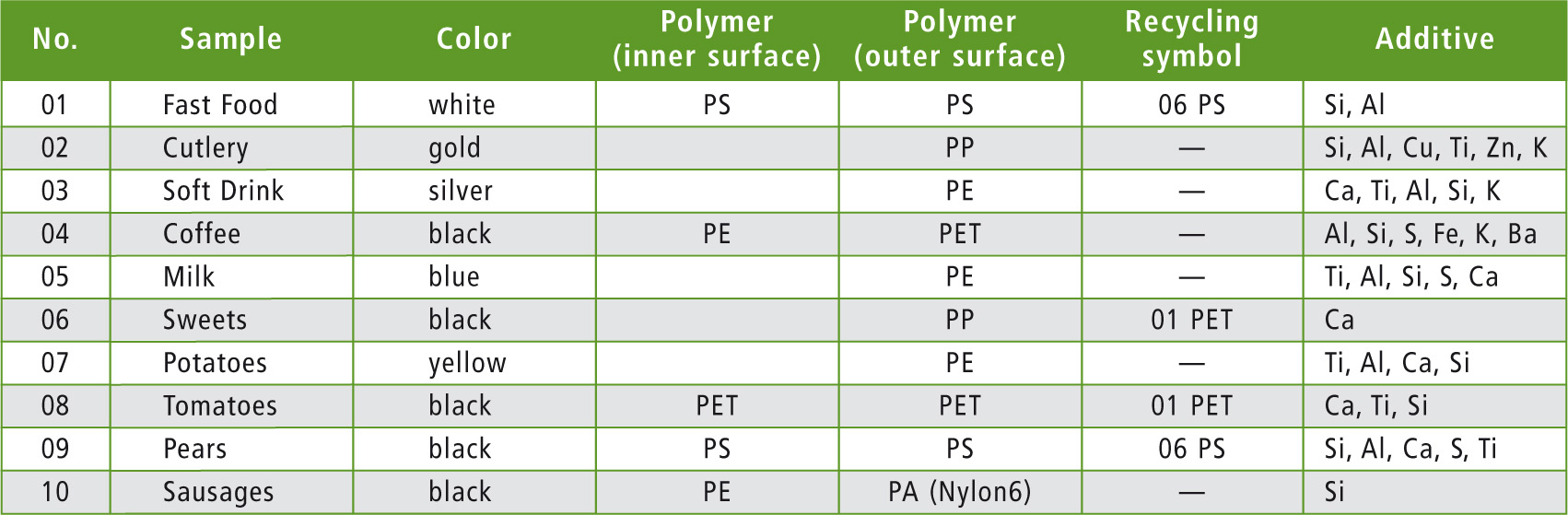 Table 1: Organic and inorganic main components of ten selected food packagings
Table 1: Organic and inorganic main components of ten selected food packagings
The most frequently used polymers are polypropylene (PP), polyethylene (PE) and polystyrene (PS). Due to its high heat resistance, PP is used for packaging hot-filled or sterilized products while PS is used mainly in disposable items due to its high susceptibility to breakage [4].
Figure 1 shows polymer composition of the investigated samples which shows great similarity to the composition of maritime microplastics occurring on a remote Maldive island, as investigated in a study by the Germany-based Bayreuth University. The microplastics particles found consisted mainly of PE, PP and PS [5].
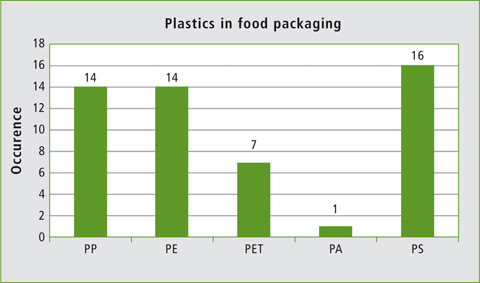 Figure 1: Occurrence of various plastics among the 43 samples investigated
Figure 1: Occurrence of various plastics among the 43 samples investigated
Quantitative analysis of heavy metals using EDX spectroscopy
EDX spectroscopy allows the identification of fillers and ROHS elements that may be present in the sample. Most samples contain larger amounts of the elements silicon, calcium, titanium and aluminum. This confirms the presence of fillers such as kaolinite (SiO2/ Al2O3) and calcite (CaCO3). The titanium is present as titanium oxide and serves not only as a white pigment but also as a stabilizer. By absorbing UV irradiation, it protects against photo-oxidation of the polymer [6].
PET is produced using an antimony trioxide catalyst [7]. For this reason, PET-containing samples were screened for antimony residues using a fast screening method specifically optimized for ROHS elements as well as for antimony and chloride. Table 2 shows the analysis results for sample 08. It contains 311.3 ppm antimony. In other PET-containing samples, antimony concentrations of several hundred ppm were found.
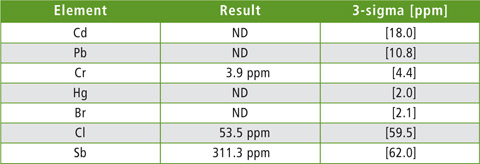 Table 2: Results of EDX screenings for ROHS elements, antimony and chlorine of sample 08. ND = not detected; 3-sigma = triple standard deviation.
Table 2: Results of EDX screenings for ROHS elements, antimony and chlorine of sample 08. ND = not detected; 3-sigma = triple standard deviation.
Sample 08 from table 1 still contains antimony residues. Many of the food packagings investigated consist of an outer PET layer and an inner PE layer. In this way, the food contained is not in direct contact with the antimony-contaminated PET layer. The examples show that a combination of FTIR and EDX spectroscopy offers access to a multitude of information on plastics and their contents. Both methods are completely non-destructive and require minimum time.
 Figure 2: PET tray for tomatoes; Sample 08
Figure 2: PET tray for tomatoes; Sample 08
MOSH/MOAH analysis using online LC-GC technology
Undesirable mineral oil residues often enter products through the use of mineral oil based printing inks which can be found in food and food packagings. This effect has been increasingly detected not only in recycled materials but also in packagings consisting of fresh raw materials. In many products, the concentration of saturated (MOSH) and aromatic (MOAH) hydrocarbons is particularly high.
For this analysis, a MOSH/ MOAH application system is now available based on LC-GC coupling and was developed in accordance with EN 16995:2016 ‘Determination of mineral oil saturated hydrocarbons (MOSH) and mineral oil aromatic hydrocarbons (MOAH) with online HPLC-GC-FID analysis; German and English version FprEN 16995:2016.’ The system consists of the following components:
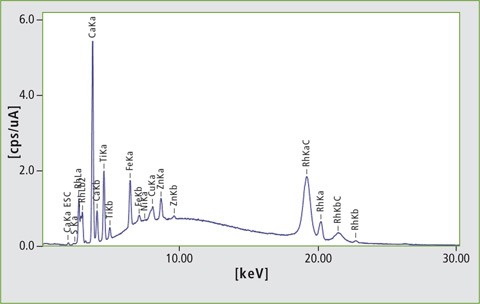 Figure 3: EDX spectrum of sample 08 (excitation energy 50 kV). The sample contains large amounts of calcium, titanium and iron.
Figure 3: EDX spectrum of sample 08 (excitation energy 50 kV). The sample contains large amounts of calcium, titanium and iron.
- Shimadzu’s LC-20ADXR pump unit with UV detector and degasser
- Shimadzu’s GC-2010 Plus with two FIDs
- CTC autosampler
- Semrau CHRONECT® LC-GC (figure 4).
 Figure 4: MOSH/MOAH application system
Figure 4: MOSH/MOAH application system
The method offers high sample throughput, reproducible results and good sensitivity. The system enables LC-GC measurements with a reproducibility comparable with conventional split/splitless injection in a gas chromatograph. Direct coupling reduces the risk of contamination in manual methods. The advantage of the system is the configuration of two FID detectors, allowing parallel determination of MOSH and MOAH within a single run and leading to characteristic chromatograms as shown in figure 5.
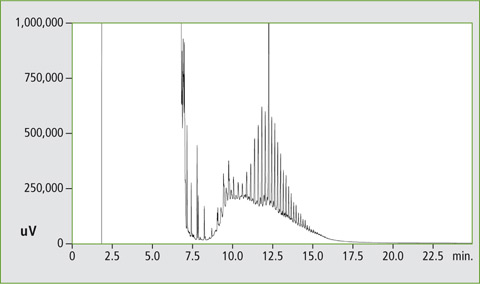
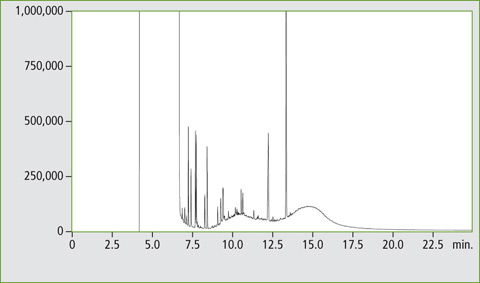 Figure 5: Chromatogram of the MOSH and MOAH fractions
Figure 5: Chromatogram of the MOSH and MOAH fractions
Conclusion
The Commission Regulation (EU) No. 10/2011 on plastic materials and articles that come into contact with food (also referred to as ‘Plastics Implementation Measure’, PIM) has been in force since 4 February 2011. As of 1 January 2016, the test conditions prescribed therein apply fully. In addition to overall migration (migration limit 60 mg/kg food), many other specific migration limits must be adhered to.
Shimadzu offers a comprehensive solution for the analysis of non-intentionally added substances (NIAS) including heavy metals (antimony trioxide) and mineral oil fractions (MOSH/MOAH).
Literature
[1] Vebraucherzentrale Hamburg: “Lebensmittelverpackungen: Unbeabsichtigt eingebrachte Stoffe”
[2] R. Lorenzini, M. Biedermann, K. Grob, D. Garbini, M. Barbanera, I. Braschi, Food Addit Contam 30 (2013) 760-770
[3] VERORDNUNG (EU) Nr. 10/2011 DER KOMMISSION vom 14. Januar 2011 über Materialien und Gegenstände aus Kunststoff, die dazu bestimmt sind, mit Lebensmitteln in Berührung zu kommen
[4] Dr. H. Saechtling: Kunststoff Taschenbuch. 31. Ausgabe, Carl Hanser Verlag, München 2013
[5] Hannes K. Imhof et al., Spatial and temporal variation of macro-, meso- and microplastic abundance on a remote coral island of the Maldives, Indian Ocean, Marine Pollution Bulletin (2017)
[6] Dr. H. Zweifel: Plastics Additives Handbook. 6. Ausgabe, Carl Hanser Verlag, München 2009
[7] DLG: www.dlg.org/verpackungsmaterial_pet.html
Further information on this article
Flyer MOSH/MOAH Analyzer