Using electrochemistrymass spectrometry to reveal tomorrow´s contaminants, today
Sparking innovation in the prediction of chemical transformation products (TPs)
Ranil C. T. Temgoua, Helmut-Schmidt-Universität/Universität der Bundeswehr (Hamburg) & Helmholtz-Zentrum Hereon, Geesthacht, Germany
Every chemical molecule has a story to tell. But what happens to these molecules after they leave the lab and enter the complex reality of nature or the human body? When a pharmaceutical product completes its therapeutic mission and is excreted, or when an agrochemical solution protects a crop and then washes into the soil, their stories are still far from over. These molecules now embark on a second, secret life: a journey of transformation. They break down, come together with other substances, and evolve into new chemical entities. How can we possibly predict their fate?


The few things we know and the many things we don’t know about the transformation of our chemicals once they perform their tasks is often called the “iceberg” problem (Figure 1a). We know the parent compound and we regulate that, but this is merely the visible tip. Below the surface, a vast unseen world of new molecules is being created: transformation products (TPs). TPs can be more persistent, more mobile, or even more toxic than the original compound, yet they remain largely unknown, undetected, and unregulated. They are the “chemical dark matter” of our environment, and scientists have struggled to find them. Understanding their journey is one of the greatest challenges in modern science.
The question that drives our research is deceptively simple: How do we predict what happens to chemicals in our environment and in our bodies without waiting months or even years for the answer? Conventional methods of studying these pathways are slow, costly, and ethically complex. Enzymatic studies using liver microsomes are variable, and environmental fate studies can take months. We have long needed a faster, more controlled, and predictive approach.
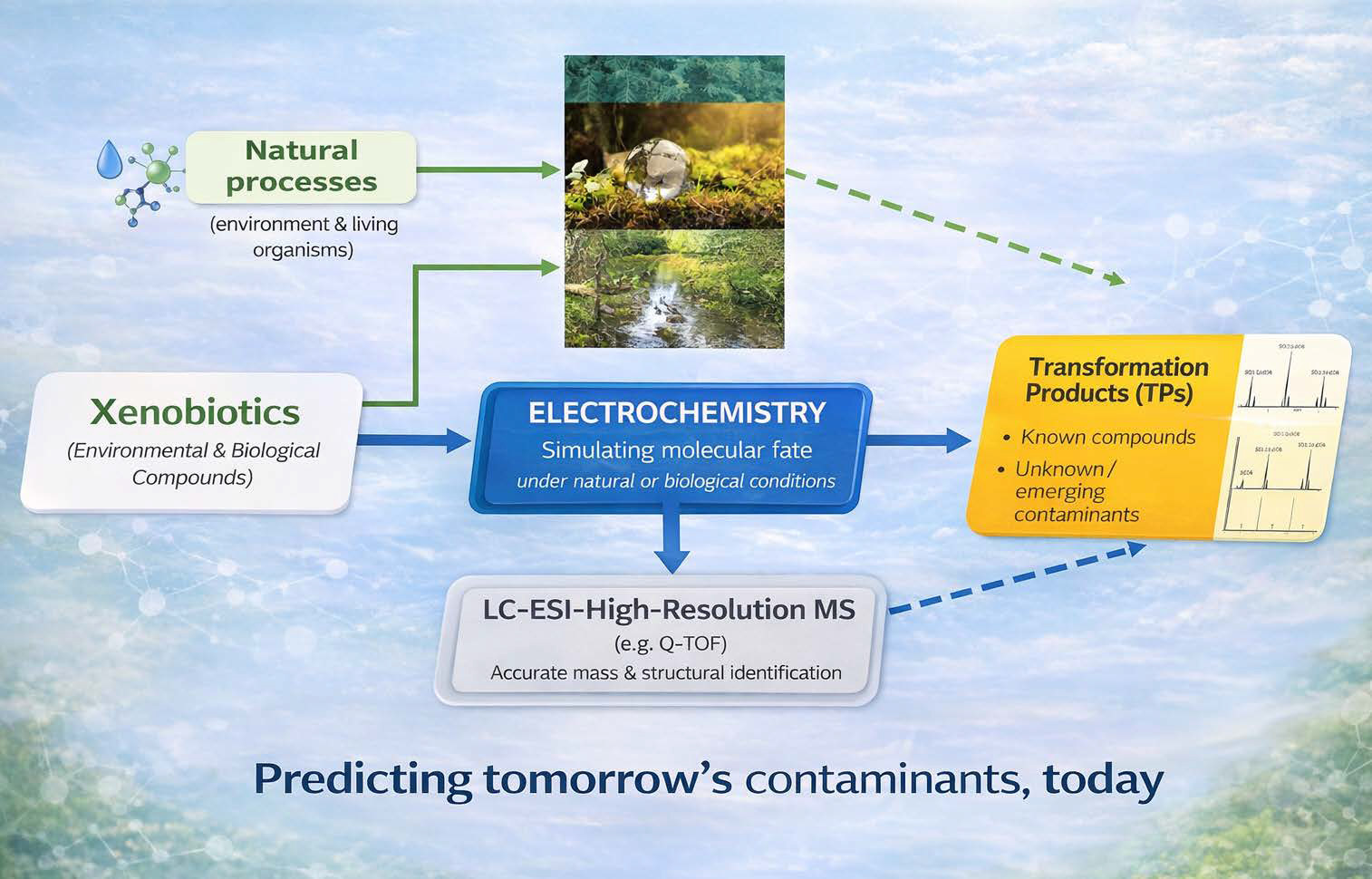
The solution lies in a powerful analytical synergy: using electrochemistry to mimic a molecule’s fate (similar to living organisms or natural systems conditions) and advanced mass spectrometry to reveal it with stunning clarity. An illustration is given in Figure 1b.
Act I: The crystal ball of electrochemistry
The breakthrough lies in a simple, yet profound, realization: Over 90 % of organic compounds are electroactive. This means they can be oxidized or reduced by applying an electrical potential. We can harness this property to simulate metabolic and environmental degradation in a “digital reactor” – an electrochemical cell.
By fine-tuning the potential in this cell, we subject a molecule to controlled redox stress, effectively accelerating its natural aging process. In a matter of minutes, we can generate the very same complex mixture of metabolites and degradation products that would form over weeks or months in nature. It’s a clean, reproducible, and ethical method that provides a high-fidelity preview of a molecule’s future. As illustrated in the diagram below (Figure 2), this electrochemical digital reactor is seamlessly integrated upstream of the mass spectrometer.
But generating these transformation products is only half the story. To truly understand their impact, we must identify them. This is where our predictive vision comes into sharp focus.
Act II: Using the LCMS-9050 Q-TOF as the ultimate decoder
The mixture flowing from our electrochemical reactor is a complex puzzle of unknown compounds. Solving it requires a detector that is not just a sensor but a master detective. This critical partner is the Shimadzu LCMS-9050 Quadrupole Time-of-Flight (Q-TOF) mass spectrometer (see Figure 3).
Its role is to interrogate every product formed with unparalleled precision. Here’s how it brings the unseen to light:
1. It sees everything: The TPs we generate are often short-lived and in trace concentrations. The outstanding sensitivity of the LCMS-9050 is therefore critical. It ensures that even the lowest-level products are not missed, capturing the most complete profile of all potential TPs.
2. It provides the “fingerprint”: Seeing is not the same as knowing. To identify a true unknown, we need its “fingerprint.” The LCMS-9050 provides stable, ultra-high resolution and highly accurate mass measurements. This isn’t just a weight; it’s a precise molecular formula, an exact identity card (e.g., C₁₂H₁₅NO₄S) and the non-negotiable first step in identifying a molecule from scratch.


3. It unlocks the structure: With this precise identity, we can use the Q-TOF’s MS/MS capability to fragment the molecule. This provides a map of its structure. This high-quality data, combined with Shimadzu’s powerful software and extensive spectral libraries, allows us to piece the puzzle together and confidently name these previously hidden compounds.

The synergy: A complete workflow for predictive science
When electrochemistry and the LCMS-9050 Q-TOF are linked in a seamless workflow, we achieve something transformative. We are no longer merely analyzing what something is; we are proactively forecasting what it could be.
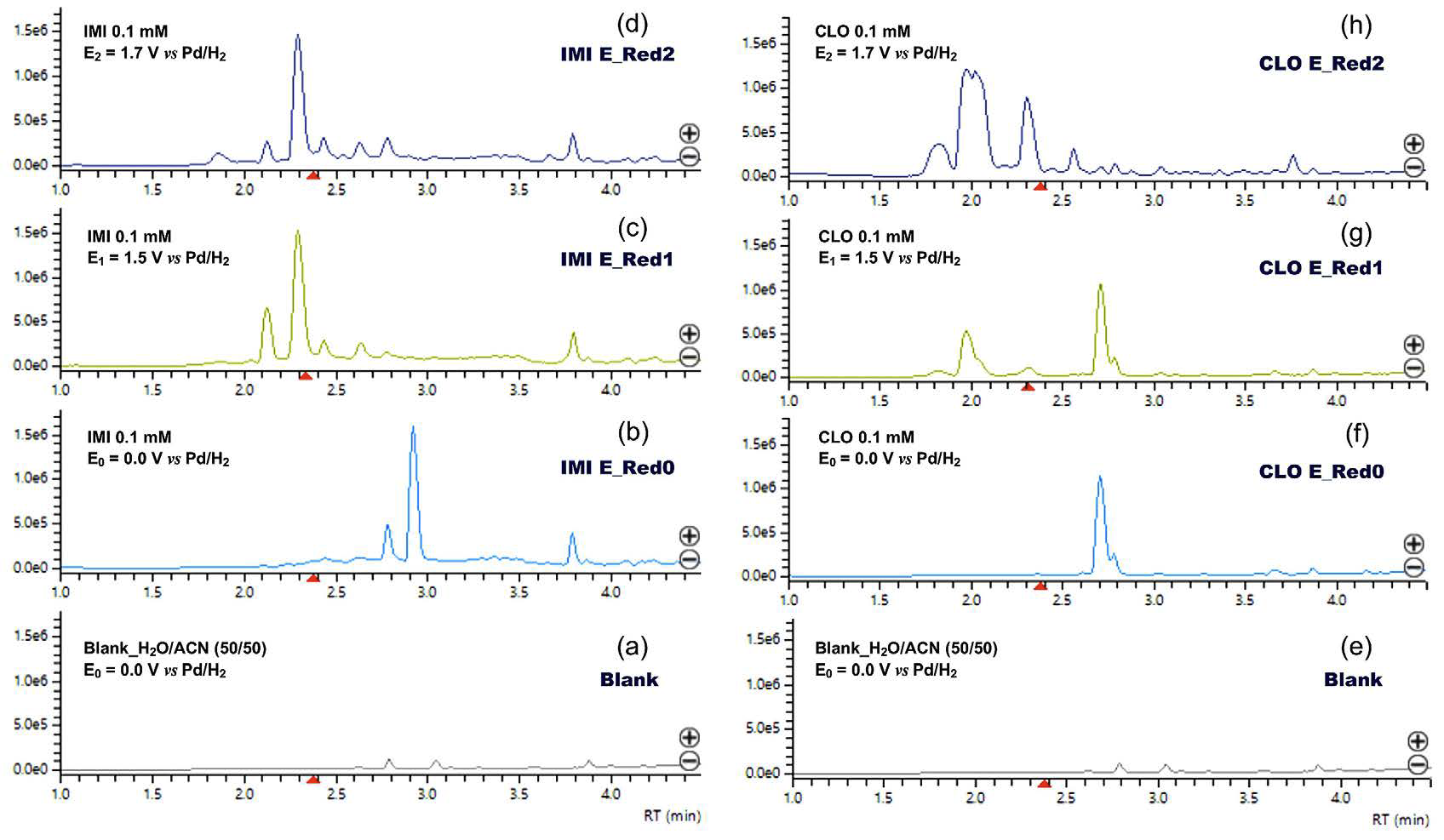
In our research, this powerful partnership allowed us to answer critical questions rapidly. A concrete example of the results, presented in Figure 4, perfectly illustrates this power: The real-time monitoring of the electrochemical degradation of two neonicotinoid pesticides (imidacloprid, IMI, and clothianidin, CLO), where the Q-TOF identified principal transformation products in less than an hour.
The results speak for themselves: While the pesticides remained stable at rest, a simple “flick of the switch” triggered their transformation. For imidacloprid, the parent molecule vanished almost instantly at –1.5 V, replaced by a fingerprint of new products. Clothianidin, more resilient, held its ground initially but finally yielded at –1.7 V, revealing its hidden degradation path. This demonstrates the surgical precision of EC-MS: We can tune the energy to break even the most stubborn molecules and map their future in minutes.
From passive observation to active design
This powerful combination of electrochemistry and mass spectrometry marks a paradigm shift. We are moving from being passive observers of chemical fate to active architects of a safer chemical future. By predicting a molecule’s journey with electrochemistry and illuminating its path with the unparalleled clarity of the Shimadzu LCMS-9050 Q-TOF, we can design better, safer, and more sustainable molecules from the very beginning.
The future of chemical analysis isn’t just about measuring what we know; it’s about discovering what we don’t. And that journey of discovery has never been faster, clearer, or more inspiring.

Bai, L., Chen, P., Xu, J. et al. (2025). Comprehensive Profiling of Chemical Constituents and Metabolites of the Angelica sinensis-Sophora flavescens Herbal Pair in Plasma and Urine via UHPLC-Q-TOF-MS Coupled With Multi-Platform Data Integration. Journal of Separation Science. 48 (6): e70201.
Baumann A., Karst U. (2010). Online electrochemistry/mass spectrometry in drug metabolism studies: principles and applications. Expert Opin Drug Metab Toxicol. 6: 715–31.
Zahn, D., Arp H.P.A. (2024). Should Transformation Products Change the Way We Manage Chemicals? Environ. Sci. Technol. 58 (18): 7710–7718.
Temgoua, R.C.T., Tonlé, I.K., Boujtita, M. (2023). Electrochemistry coupled with mass spectrometry for the prediction of the environmental fate and elucidation of the degradation mechanisms of pesticides: current status and future prospects. Environ. Sci.: Processes Impacts. 25: 340–350.