How green is your solvent –
and how do you know
GreenSOL is the first in-depth solvent greenness guide for analytical chemists
Elia Psillakis, Technical University of Crete, Greece
Eugenio Alladio, University of Torino, Italy
Alice Mondello, University of Messina, Italy
Paolo Oliveri, University of Genova, Italy
Francisco Pena-Pereira, University of Vigo, Spain
Analytical laboratories are shifting toward greener practices. As a result, solvents are often assessed and selected for their lower toxicity and safer profiles during laboratory handling. However, this narrow lens of assessment can create an illusion of greenness, where risks are simply relocated to another stage of the solvent’s life cycle. A “greener” solvent may, in fact, be highly energy-intensive to manufacture or present a major challenge for waste treatment. True progress in green analytical chemistry requires looking deeper into the complete life cycle of solvents.
Solvents and the move toward more sustainable practices
Solvents are indispensable in analytical chemistry, directly impacting the outcome and reliability of the analytical results. While their consumption in analytical sciences is a fraction of industrial use, the cumulative volumes are still staggering: Liquid chromatography alone uses an estimated 150 million liters annually. Current trends, aligned with the principles of green analytical chemistry and green sample preparation, advocate the use of solvents that minimize environmental, health and safety (EHS) impacts as a critical strategy for mitigating the environmental footprint of analytical procedures and safeguarding laboratory personnel. In addition, initiatives to reduce solvent consumption (e.g., through miniaturization and the implementation of recovery, reuse and recycling strategies) support the broader goals of circular analytical chemistry [1] and advance sustainability in the field [2]. These developments are further propelled by increasingly stringent regulations on solvent usage and disposal, alongside a growing emphasis on greener laboratory practices.
The GreenSOL solvent life cycle story
For decades, solvent selection in analytical chemistry has lacked a clear, evidence-based guide tailored to its distinct needs. While solvent guides existed, they were designed primarily for industrial synthesis rather than analytical applications. To address this gap, a European team of researchers – guided by the principles of green and circular analytical chemistry – has developed GreenSOL, the first comprehensive solvent selection guide designed specifically for analytical applications.
To assess greenness, GreenSOL adopts a life cycle approach and evaluates each solvent across its production, laboratory use and waste phases. First published in TrAC-Trends in Analytical Chemistry GreenSOL is more than a high-impact scientific paper, it’s a practical guide, accompanied by a free, interactive, web-based tool empowering analysts to make informed solvent choices [3]. GreenSOL evaluates 49 common and niche solvents, selected to ensure broad coverage of chemical classes and physicochemical properties relevant to analytical workflows. In addition, nine deuterated solvents are included: These have previously been overlooked in solvent guides.

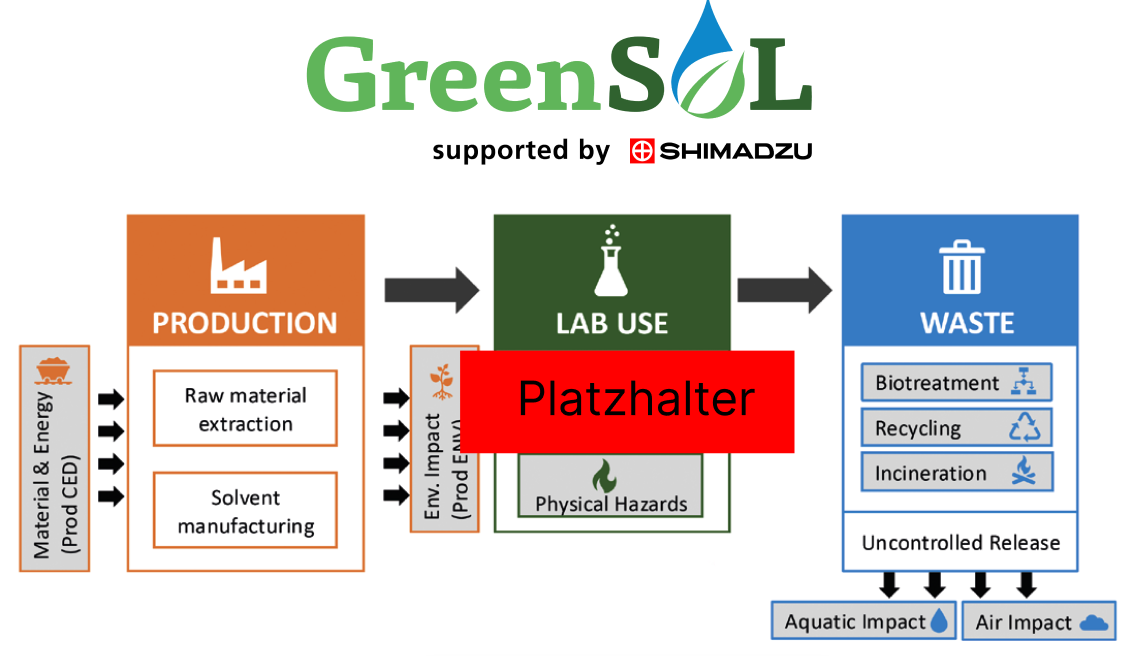
The three phases of assessment: Production, use and waste
A large part of GreenSOL’s uniqueness lies in its three-phase life cycle evaluation system (Figure 1). Each of the three phases depends on multiple attributes and has subcategories. GreenSOL ensures that a single solvent characteristic (e.g., high volatility) is appropriately accounted for within each phase, while preventing multiple penalties for the same characteristic.
In GreenSOL, individual subcategories are assessed on a scale of 1–10 (1 = least favorable, 10 = most recommended) and then combined to generate a composite score for each phase. Composite phase scores are helpful for a quick overview, but the real strength of the guide lies in the detailed subcategory breakdown. GreenSOL presents this comprehensive perspective, empowering analysts to prioritize criteria based on their specific application and operational context.
The three phases used in GreenSOL are:
Production Phase: Quantifies the upstream environmental burden using a cradle-to-gate life cycle assessment (LCA) with key subcategories including cumulative energy demand and environmental impact during production.
Use Phase: Assesses immediate laboratory risks by considering physical and health hazards.
Waste Phase: Evaluates the downstream fate of the solvent through two lenses:
• Environmental impact: The potential harm from uncontrolled release into aquatic species and the atmosphere.
• Treatment potential: The effectiveness of controlled disposal via the three most common methods: biotreatment, recycling and incineration
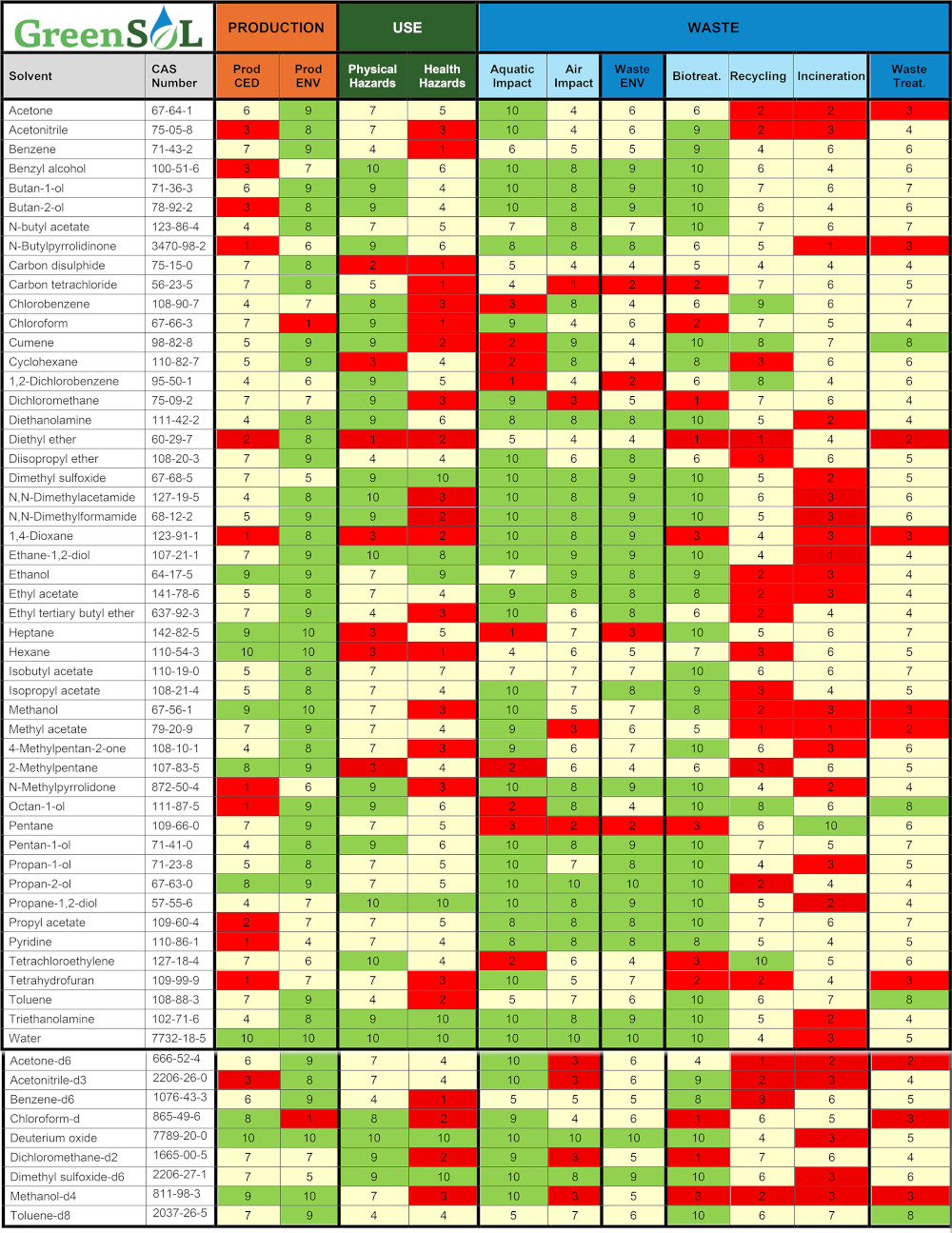
The detailed results of the solvent assessments are presented in Table 1. A color-coding scheme is applied to scores within each category and subcategory to facilitate the visual comparison of solvent performance. The colors used are: red (1 ≤ score ≤ 3), yellow (4 ≤ score ≤ 7) and green (8 ≤ score ≤ 10).
In many ways, GreenSOL is full of surprises, precisely because it exposes an uncomfortable reality: Solvent greenness is a complex, multi-layered compromise rather than a simple label. Our results powerfully reinforce the need to evaluate the entire life cycle. For instance, hexane’s lab use score was, as expected, very poor. However, its proposed “greener” replacements reveal their own trade-offs. While pentane reduces laboratory risks, it has a substantial environmental impact in the event of an uncontrolled release. Meanwhile, heptane lowers health risks but retains high physical hazards and poses a greater risk to aquatic species.
A digital tool for evidence-based decision-making
Understanding that a static table in a paper has limited daily utility, the team developed an interactive, free web application with the assistance of Shimadzu (https://greensol.tuc.gr/). This is where GreenSOL really comes to life, transforming it from an academic exercise into a decision-support system for routine lab work, method development and educational training. Analysts can filter solvents based on the physicochemical properties they need (e.g., boiling point, polarity or water solubility) and instantly see how the candidates rank in terms of greenness. They can also compare solvents within the same chemical family, find viable alternatives or create custom 2D and 3D plots to visualize trade-offs between production, use and waste scores.
Another actionable step labs can take to reduce their impact is to set and enforce green procurement and safety standards using GreenSOL’s scores. This can be achieved by excluding any solvent with a critically poor score in a non-negotiable category, such as health hazards. For commonly used solvents that are difficult to replace, analysts can use subcategory scores to select the option with the lowest overall burden, making informed trade-offs between, for example, production energy demand and waste treatment viability.
GreenSOL aligns with a powerful shift in the field: the move toward circular analytical chemistry. This philosophy emphasizes minimizing waste, recovering resources and designing inherently sustainable processes. By highlighting the waste phase, labs are encouraged to think about solvent recovery and reuse. A waste treatment strategy can be built by using waste-phase data. For solvents with high recycling scores, implementing even simple on-site recovery can turn a costly lab into a circular asset.
Using the truth constructively
GreenSOL highlights that true solvent greenness is not about finding a perfect choice but about navigating inevitable trade-offs with responsibility and insight. Transparency is essential to prevent burden-shifting from one life cycle stage to another. A reliable guide must reveal these compromises rather than conceal them: Focusing solely on toxicity may ignore a high energy burden, while optimizing only for production could create a downstream waste problem. This holistic approach raises the question: Are we solving one issue by creating another elsewhere in the solvent’s life? This shift to life-cycle thinking is the fundamental lesson.
The search for a perfectly “green” solvent is like chasing a myth. No single solvent satisfies all criteria across its entire life cycle. Instead, our practical goal should be to make informed choices that minimize overall enviornmental and health impacts. In fact, the biggest gains from our considerable efforts to reduce solvent impacts are achieved through reductions in solvent use and method optimization.
The ultimate message is clear: The greenest solvent is the one you do not use. But when a solvent is necessary, selecting the greenest option is now a guided, evidence-based decision. GreenSOL provides the missing map for this journey, marking the path to more sustainable laboratories and a better future.
[1] Psillakis E., Pena-Pereira F. (2024). The twelve goals of circular analytical chemistry. TrAC Trends in Analytical Chemistry. 175: 117686–117686. https://doi.org/10.1098/rsos.21200.
[2] Psillakis E. (2025). Towards sustainable analytical chemistry. TrAC Trends in Analytical Chemistry. 191: 118371. https://doi.org/10.1016/j.trac.2025.118371.
[3] Stampolaki, E., Mondello, A., Alladio, E., Oliveri, P., Mazzoleni, A., Pena-Pereira, F., Psillakis, E. (2026). GreenSOL: Green solvent guide for analytical chemistry based on production-to-end-of-life assessment. TrAC Trends in Analytical Chemistry. 194: 118531. https://doi.org/10.1016/j.trac.2025.118531.
[1] Psillakis E., Pena-Pereira F. (2024). The twelve goals of circular analytical chemistry. TrAC Trends in Analytical Chemistry. 175: 117686–117686. https://doi.org/10.1098/rsos.21200.
[2] Psillakis E. (2025). Towards sustainable analytical chemistry. TrAC Trends in Analytical Chemistry. 191: 118371. https://doi.org/10.1016/j.trac.2025.118371.
[3] Stampolaki, E., Mondello, A., Alladio, E., Oliveri, P., Mazzoleni, A., Pena-Pereira, F., Psillakis, E. (2026). GreenSOL: Green solvent guide for analytical chemistry based on production-to-end-of-life assessment. TrAC Trends in Analytical Chemistry. 194: 118531. https://doi.org/10.1016/j.trac.2025.118531.
Elefteria Psillakis: Conceptualisation, Methodology, Use and Waste Phases Data Collection and Calculations
Alice Mondello: Production Phase Data Collection and Calculations (LCA studies)
Eugenio Alladio and Paolo Oliveri: Web app development
Francisco Pena-Pereira: Validation